Can Henna Be Used For Makeup
Prophylactic and Regulatory Data
FDA has received reports of adverse reactions to some "decal," henna, and "blackness henna" temporary tattoos. Here is information about the rubber of these products and how they are regulated.
- "Decal" Temporary Tattoos
- Henna, or Mehndi, and "Black Henna"
- Finding out What's in a Temporary Tattoo or Henna/Mehndi Product
- FDA'due south Say-so over Color Additives in Cosmetics
- FDA'southward Say-so over Other Corrective Ingredients
- FDA Enforcement Action
- How to Report a Reaction to a Temporary Tattoo or Other Cosmetic
- More Resources on Temporary Tattoos:
- Related Resource:
"Decal" Temporary Tattoos
Decal temporary tattoos are used to decorate any part of the body, including areas of the face and around the eyes, and may last for a 24-hour interval or upwardly to a calendar week or more. They are especially pop with children and at Halloween.
There are two kinds of decal tattoos:
- Some are images attached to a removable bankroll. The decal prototype is removed from the backing by wetting, and the image is then practical directly to the skin.
- Others have a backing that adheres to the skin, creating a partial or complete barrier between the skin and the dyes used in the image.
The difference is important, because not all dyes are known to be rubber for use on the pare. While an adhesive backing may protect the skin from unapproved colors, there may be other ingredients on or in the decal to help the image attach amend either to the backing or to the pare that may cause problems for some people.
FDA has received reports of reactions to some decal-type temporary tattoos. Before using a temporary tattoo on your face, it may be a expert idea to endeavor it on a less conspicuous role of your torso.
Henna, or Mehndi, and "Black Henna"
Henna, a coloring made from a constitute, is approved only for employ as a hair dye. Information technology is not approved for straight application to the skin, every bit in the body-decorating process known as mehndi. This unapproved employ of a color additive makes these products adulterated. Information technology is unlawful, for instance, to introduce an adulterated cosmetic into interstate commerce.
Considering henna typically produces a brown, orange-dark-brown, or crimson-brownish tint, other ingredients must be added to produce other colors, such every bit those marketed as "black henna" and "blue henna." Even brownish shades of products marketed every bit henna may contain other ingredients intended to make them darker or make the stain last longer on the skin.
The extra ingredient used to blacken henna is often a coal-tar pilus dye containing p-phenylenediamine (PPD), an ingredient that can crusade dangerous skin reactions in some people. That's the reason pilus dyes have a caution statement and instructions to do a "patch test" on a small area of the skin earlier using them. Sometimes, the creative person may use a PPD-containing hair dye alone. Either way, there's no telling who will be affected. Past law, PPD is not permitted in cosmetics intended to be practical to the skin.
FDA has received reports of injuries to the peel from products marketed as henna and products marketed as "black henna." For more data on Henna, see the consumer update: Temporary Tattoos May Put You at Risk.
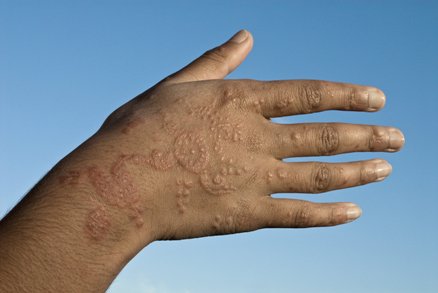
Allergic reaction on a man's hand. J. Cole/Photo Researchers.
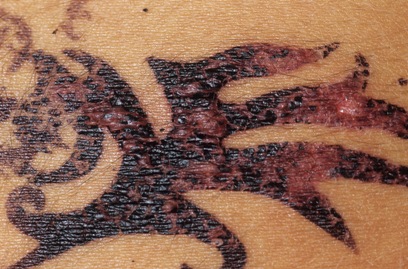
Allergic reaction on a 14-twelvemonth-sometime girl. Dr. P. Marazzi/Photo Researchers.
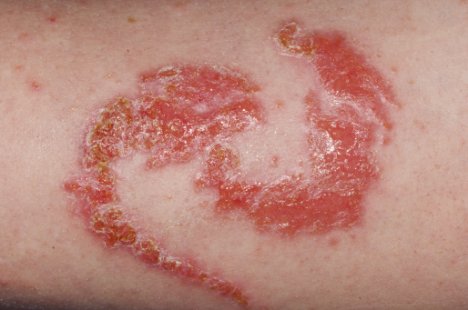
Allergic reaction on an arm. Dr. P. Marazzi/Photograph Researchers.
Finding out What's in a Temporary Tattoo or Henna/Mehndi Production
Cosmetics that are sold on a retail basis to consumers must have their ingredients listed on the characterization. Without such an ingredient proclamation, they are considered misbranded, and therefore it is unlawful to innovate them into interstate commerce. FDA requires the ingredient declaration under the authority of the Fair Packaging and Labeling Act (FPLA).
Because the FPLA does non utilise to corrective samples and products used simply by professionals--for example, for application at a salon, or a booth at a off-white or boardwalk--the requirement for an ingredient declaration does non apply to these products.
FDA'south Authority over Color Additives in Cosmetics
By law, all color additives used in cosmetics must be approved by FDA for their intended uses, with the exception of coal tar colors intended for use in pilus dyes. In add-on, some colour additives must not be used unless FDA has certified that the batch meets the regulatory requirements for composition and purity. Cosmetics, including temporary tattoo products, that do not comply with restrictions on color additives are considered adulterated, and it is unlawful to introduce them into interstate commerce. To learn more, see Colour Additives and Cosmetics, and, for information on how color additives are approved, Color Additive Petitions.
FDA's Authority over Other Cosmetic Ingredients
Cosmetics must be rubber when consumers utilise them following directions on the label, or in the customary or expected way. Except for colour additives, the law does non require cosmetic products and ingredients to have FDA approval before they are marketed. In addition, firms are not required to report their safety information, including complaints.
For a list of ingredients that are prohibited or restricted in cosmetics, run across "Prohibited and Restricted Ingredients."
For a list of colour additives allowed in cosmetics, how they are allowed to be used, and links to their regulations, see "Color Additives Permitted for Use in Cosmetics."
FDA Enforcement Action
FDA can accept action confronting cosmetics on the market that don't comply with the law. For case, nosotros tin effect Import Alerts and Alert Letters.
An Import Alert allows FDA to detain products that violate or appear to violate the Federal Food, Drug, and Corrective Act. We have two Import Alerts in effect for temporary tattoos. However, considering not all shipments of imported cosmetics are inspected, it's notwithstanding possible for some unsafe or mislabeled products to be imported into this country.
- An Import Alarm is in effect for several strange-made decal temporary tattoos because they incorporate colors non permitted for employ in cosmetics applied to the skin, or they don't have the required ingredient list on the label, or they are labeled every bit "FDA approved."
- An Import Alert is in effect for henna intended for use on the skin considering information technology is an unapproved use of the color additive.
FDA issues Warning Letters to let companies know that they accept violated the police and to tell them what corrective action they need to take. We have issued a Warning Alphabetic character to a company marketing "black henna" products:
- Warning Letters to Industry on Corrective-Related Issues
It is of import to annotation that the practice of tattooing is mostly regulated by state and/or local officials, and that the FDA is not typically involved in such enforcement. While states have jurisdiction over professional practices such as tattooing and cosmetology, that oversight differs from state to state. Some states have laws and regulations for temporary tattooing, while others don't. So, depending on where you are, it'southward possible no i is checking to brand certain the artist is following safe practices or even knows what may exist harmful to consumers.
How to Study a Reaction to a Temporary Tattoo or Other Corrective
It's important for consumers and health care providers to study bug with cosmetics to FDA. This information helps FDA find out which products are causing bug, and what kinds of problems.
You can report a problem with a cosmetic to FDA in either of these ways:
- Contact MedWatch, FDA'due south trouble-reporting program, on the Web or at 1-800-332-1088
, or file a MedWatch Voluntary report online.
- Contact the consumer complaint coordinator in your expanse.
To learn more, run into Agin Event Reporting.
Related Resources
- Consumer Update: Temporary Tattoos May Put You at Hazard
- Temporary Tattoos: Raising Consumer Sensation of Safety: FDA Webinar, May 13, 2014
- Warning Letter Issued to Black Henna Ink, Inc.
- Import Alert #53-19: Detention Without Physical Test of Henna Based Skin Color
- Lucky 13 Tips for a Safe Halloween
- Novelty Makeup: Special for Halloween
- Tattoos and Permanent Makeup
- FDA Dominance Over Cosmetics
- FDA Recall Policy for Cosmetics
- Tattoos & Permanent Makeup: Quick Guide (PDF: 536 KB)
- Los Tatuajes y el Maquillaje Permanente: Una Guía (PDF: 522KB)
Can Henna Be Used For Makeup,
Source: https://www.fda.gov/cosmetics/cosmetic-products/temporary-tattoos-hennamehndi-and-black-henna-fact-sheet
Posted by: lujancoldingaze.blogspot.com


0 Response to "Can Henna Be Used For Makeup"
Post a Comment