What Is The Makeup Of Carbohydrates
Learning Outcomes
- Distinguish between monosaccharides, disaccharides, and polysaccharides
- Identify several major functions of carbohydrates
Most people are familiar with carbohydrates, one type of macromolecule, especially when it comes to what nosotros eat. To lose weight, some individuals attach to "low-carb" diets. Athletes, in contrast, often "carb-load" before important competitions to ensure that they take enough free energy to compete at a high level. Carbohydrates are, in fact, an essential part of our diet; grains, fruits, and vegetables are all natural sources of carbohydrates. Carbohydrates provide free energy to the body, particularly through glucose, a unproblematic saccharide that is a component of starch and an ingredient in many staple foods. Carbohydrates also take other important functions in humans, animals, and plants.
Carbohydrates can exist represented by the stoichiometric formula (CH2O)northward, where n is the number of carbons in the molecule. In other words, the ratio of carbon to hydrogen to oxygen is 1:ii:1 in carbohydrate molecules. This formula also explains the origin of the term "carbohydrate": the components are carbon ("carbo") and the components of h2o (hence, "hydrate"). Carbohydrates are classified into three subtypes: monosaccharides, disaccharides, and polysaccharides.
Monosaccharides
Monosaccharides (mono– = "one"; sacchar– = "sweet") are simple sugars, the most common of which is glucose. In monosaccharides, the number of carbons ordinarily ranges from iii to seven. Most monosaccharide names end with the suffix –ose. If the sugar has an aldehyde group (the functional group with the structure R-CHO), information technology is known as an aldose, and if it has a ketone grouping (the functional grouping with the structure RC(=O)R′), it is known as a ketose. Depending on the number of carbons in the carbohydrate, they also may be known as trioses (three carbons), pentoses (five carbons), and or hexoses (six carbons). See Figure 1 for an illustration of the monosaccharides.

Figure 1. Monosaccharides are classified based on the position of their carbonyl group and the number of carbons in the backbone. Aldoses have a carbonyl group (indicated in green) at the cease of the carbon chain, and ketoses have a carbonyl grouping in the heart of the carbon chain. Trioses, pentoses, and hexoses have three, five, and half dozen carbon backbones, respectively.
The chemical formula for glucose is Chalf dozenH12Osix. In humans, glucose is an of import source of energy. During cellular respiration, energy is released from glucose, and that energy is used to help make adenosine triphosphate (ATP). Plants synthesize glucose using carbon dioxide and water, and glucose in plough is used for energy requirements for the found. Backlog glucose is often stored equally starch that is catabolized (the breakdown of larger molecules by cells) by humans and other animals that feed on plants.
Galactose and fructose are other common monosaccharides — galactose is institute in milk sugars and fructose is found in fruit sugars. Although glucose, galactose, and fructose all accept the same chemic formula (CviH12Ovi), they differ structurally and chemically (and are known as isomers) because of the different arrangement of functional groups around the asymmetric carbon; all of these monosaccharides take more than than one asymmetric carbon (Figure 2).
Do Question
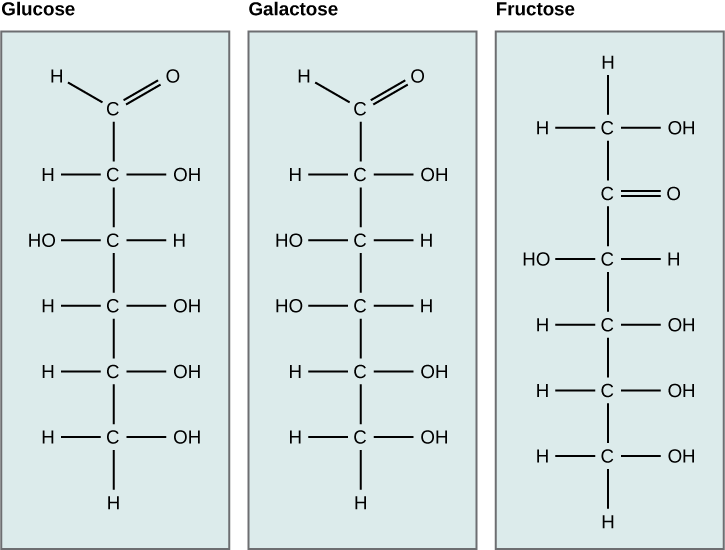
Effigy ii. Glucose, galactose, and fructose are all hexoses. They are structural isomers, meaning they have the same chemical formula (C6H12O6) but a dissimilar arrangement of atoms.
What kind of sugars are these, aldose or ketose?
Bear witness Answer
Glucose and galactose are aldoses. Fructose is a ketose.
Monosaccharides tin can be as a linear chain or as ring-shaped molecules; in aqueous solutions they are normally institute in ring forms (Effigy three). Glucose in a band course tin can have ii different arrangements of the hydroxyl group (−OH) around the anomeric carbon (carbon 1 that becomes asymmetric in the process of ring formation). If the hydroxyl group is beneath carbon number i in the saccharide, it is said to exist in the alpha (α) position, and if it is above the aeroplane, it is said to be in the beta (β) position.

Figure three. Five and six carbon monosaccharides exist in equilibrium between linear and ring forms. When the band forms, the side chain it closes on is locked into an α or β position. Fructose and ribose as well grade rings, although they class five-membered rings as opposed to the six-membered ring of glucose.
Disaccharides
Disaccharides (di– = "two") class when two monosaccharides undergo a aridity reaction (also known as a condensation reaction or dehydration synthesis). During this procedure, the hydroxyl group of ane monosaccharide combines with the hydrogen of another monosaccharide, releasing a molecule of h2o and forming a covalent bond. A covalent bond formed betwixt a carbohydrate molecule and some other molecule (in this case, between two monosaccharides) is known as a glycosidic bond (Figure 4). Glycosidic bonds (as well called glycosidic linkages) can be of the alpha or the beta type. An alpha bond is formed when the OH group on the carbon-1 of the get-go glucose is below the ring plane, and a beta bail is formed when the OH group on the carbon-i is to a higher place the band plane.

Figure 4. Sucrose is formed when a monomer of glucose and a monomer of fructose are joined in a dehydration reaction to form a glycosidic bail. In the procedure, a h2o molecule is lost. Past convention, the carbon atoms in a monosaccharide are numbered from the terminal carbon closest to the carbonyl group. In sucrose, a glycosidic linkage is formed between carbon ane in glucose and carbon ii in fructose.
Common disaccharides include lactose, maltose, and sucrose (Figure 5). Lactose is a disaccharide consisting of the monomers glucose and galactose. It is found naturally in milk. Maltose, or malt sugar, is a disaccharide formed by a aridity reaction between two glucose molecules. The most common disaccharide is sucrose, or table sugar, which is equanimous of the monomers glucose and fructose.

Effigy 5. Common disaccharides include maltose (grain sugar), lactose (milk saccharide), and sucrose (table sugar).
Polysaccharides
A long chain of monosaccharides linked by glycosidic bonds is known as apolysaccharide (poly– = "many"). The chain may exist branched or unbranched, and it may contain dissimilar types of monosaccharides. The molecular weight may exist 100,000 daltons or more depending on the number of monomers joined. Starch, glycogen, cellulose, and chitin are principal examples of polysaccharides.
Starch is the stored course of sugars in plants and is fabricated up of a mixture of amylose and amylopectin (both polymers of glucose). Plants are able to synthesize glucose, and the excess glucose, beyond the plant'due south firsthand energy needs, is stored as starch in different plant parts, including roots and seeds. The starch in the seeds provides nutrient for the embryo every bit it germinates and tin also act as a source of food for humans and animals. The starch that is consumed past humans is broken down by enzymes, such as salivary amylases, into smaller molecules, such equally maltose and glucose. The cells can then blot the glucose.
Starch is made up of glucose monomers that are joined byα ane-iv or α 1-vi glycosidic bonds. The numbers one-four and 1-6 refer to the carbon number of the two residues that have joined to grade the bond. Every bit illustrated in Figure 6, amylose is starch formed by unbranched chains of glucose monomers (only α 1-4 linkages), whereas amylopectin is a branched polysaccharide (α 1-six linkages at the co-operative points).

Figure 6. Amylose and amylopectin are 2 different forms of starch. Amylose is composed of unbranched bondage of glucose monomers continued by α i,4 glycosidic linkages. Amylopectin is composed of branched chains of glucose monomers continued by α 1,4 and α 1,6 glycosidic linkages. Because of the way the subunits are joined, the glucose chains take a helical structure. Glycogen (not shown) is like in structure to amylopectin only more highly branched.
Glycogen is the storage course of glucose in humans and other vertebrates and is made up of monomers of glucose. Glycogen is the animate being equivalent of starch and is a highly branched molecule ordinarily stored in liver and muscle cells. Whenever claret glucose levels decrease, glycogen is broken down to release glucose in a process known as glycogenolysis.
Cellulose is the most arable natural biopolymer. The jail cell wall of plants is mostly made of cellulose; this provides structural support to the cell. Wood and paper are mostly cellulosic in nature. Cellulose is made up of glucose monomers that are linked pastβ one-4 glycosidic bonds (Effigy 7).

Figure vii. In cellulose, glucose monomers are linked in unbranched chains by β one-four glycosidic linkages. Because of the way the glucose subunits are joined, every glucose monomer is flipped relative to the next one resulting in a linear, fibrous structure.
Every bit shown in Figure 7, every other glucose monomer in cellulose is flipped over, and the monomers are packed tightly equally extended long chains. This gives cellulose its rigidity and loftier tensile strength—which is then important to plant cells. While theβ 1-4 linkage cannot exist broken downward by human digestive enzymes, herbivores such equally cows, koalas, buffalos, and horses are able, with the help of the specialized flora in their stomach, to digest plant material that is rich in cellulose and use it as a food source. In these animals, sure species of leaner and protists reside in the rumen (part of the digestive arrangement of herbivores) and secrete the enzyme cellulase. The appendix of grazing animals also contains bacteria that digest cellulose, giving information technology an important role in the digestive systems of ruminants. Cellulases can suspension downwardly cellulose into glucose monomers that can be used as an energy source by the animal. Termites are also able to intermission down cellulose because of the presence of other organisms in their bodies that secrete cellulases.
Effigy viii. Insects have a difficult outer exoskeleton made of chitin, a blazon of polysaccharide.
Carbohydrates serve various functions in different animals. Arthropods (insects, crustaceans, and others) have an outer skeleton, called the exoskeleton, which protects their internal torso parts (as seen in the bee in Figure 8).
This exoskeleton is made of the biological macromolecule chitin, which is a polysaccharide-containing nitrogen. It is made of repeating units of Due north-acetyl-β-d-glucosamine, a modified sugar. Chitin is also a major component of fungal cell walls; fungi are neither animals nor plants and form a kingdom of their own in the domain Eukarya.
In Summary: Structure and Function of Carbohydrates
Carbohydrates are a group of macromolecules that are a vital energy source for the cell and provide structural support to institute cells, fungi, and all of the arthropods that include lobsters, crabs, shrimp, insects, and spiders. Carbohydrates are classified as monosaccharides, disaccharides, and polysaccharides depending on the number of monomers in the molecule. Monosaccharides are linked by glycosidic bonds that are formed as a upshot of dehydration reactions, forming disaccharides and polysaccharides with the elimination of a h2o molecule for each bond formed. Glucose, galactose, and fructose are mutual monosaccharides, whereas mutual disaccharides include lactose, maltose, and sucrose. Starch and glycogen, examples of polysaccharides, are the storage forms of glucose in plants and animals, respectively. The long polysaccharide chains may be branched or unbranched. Cellulose is an instance of an unbranched polysaccharide, whereas amylopectin, a constituent of starch, is a highly branched molecule. Storage of glucose, in the form of polymers similar starch or glycogen, makes information technology slightly less accessible for metabolism; however, this prevents it from leaking out of the cell or creating a loftier osmotic pressure that could cause excessive water uptake by the cell.
Effort It
Contribute!
Did you accept an idea for improving this content? Nosotros'd love your input.
Improve this pageLearn More than
Source: https://courses.lumenlearning.com/wm-biology1/chapter/reading-types-of-carbohydrates/
Posted by: lujancoldingaze.blogspot.com

0 Response to "What Is The Makeup Of Carbohydrates"
Post a Comment